
However, molecular structure is actually three-dimensional, and it is important to be able to describe molecular bonds in terms of their distances, angles, and relative arrangements in space ( ). Thus far, we have used two-dimensional Lewis structures to represent molecules. Assess the polarity of a molecule based on its bonding and structure.Explain the concepts of polar covalent bonds and molecular polarity.Predict the structures of small molecules using valence shell electron pair repulsion (VSEPR) theory.Appendix L: Standard Electrode (Half-Cell) Potentialsīy the end of this section, you will be able to:. 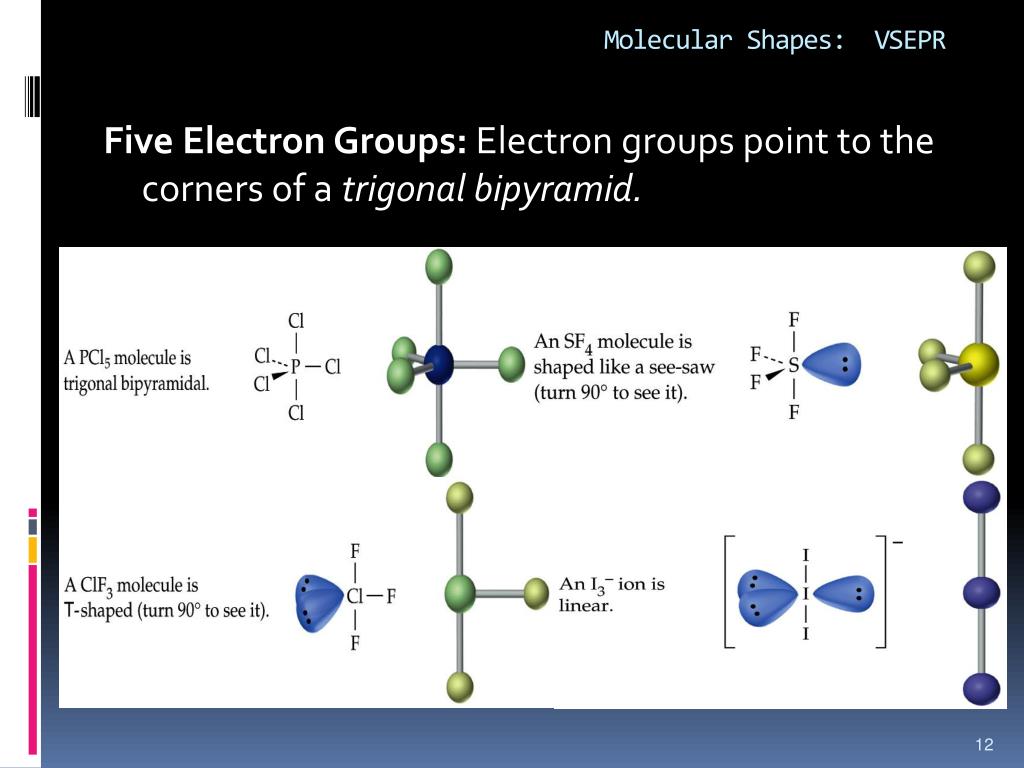
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
